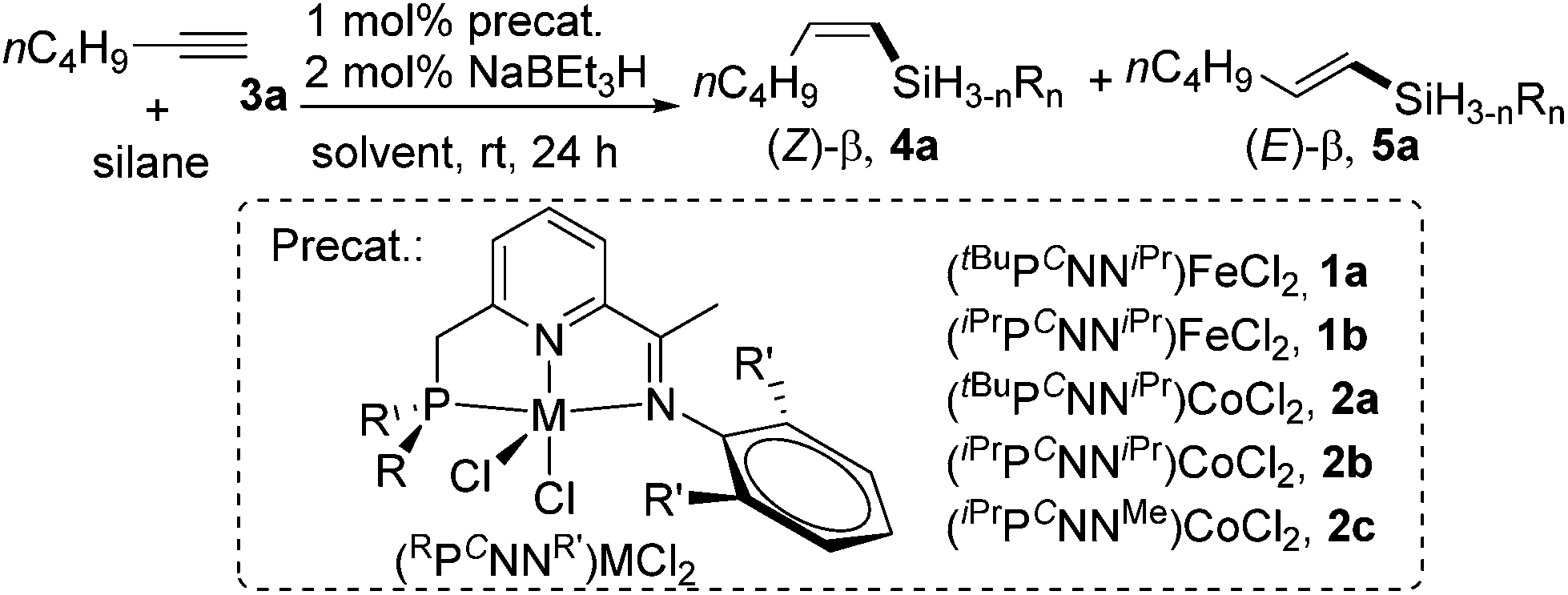
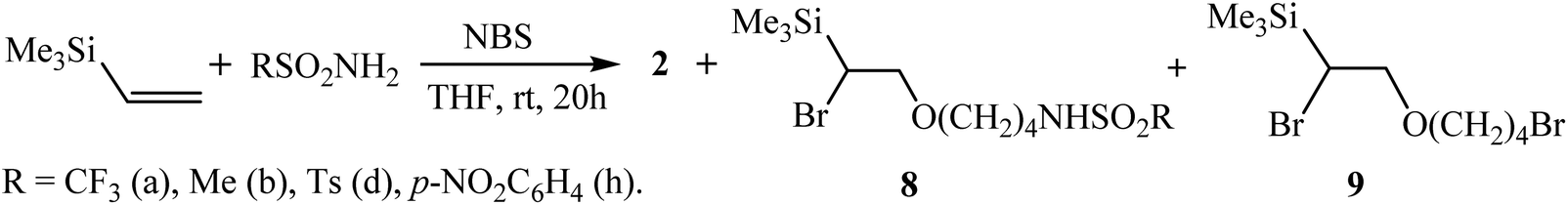
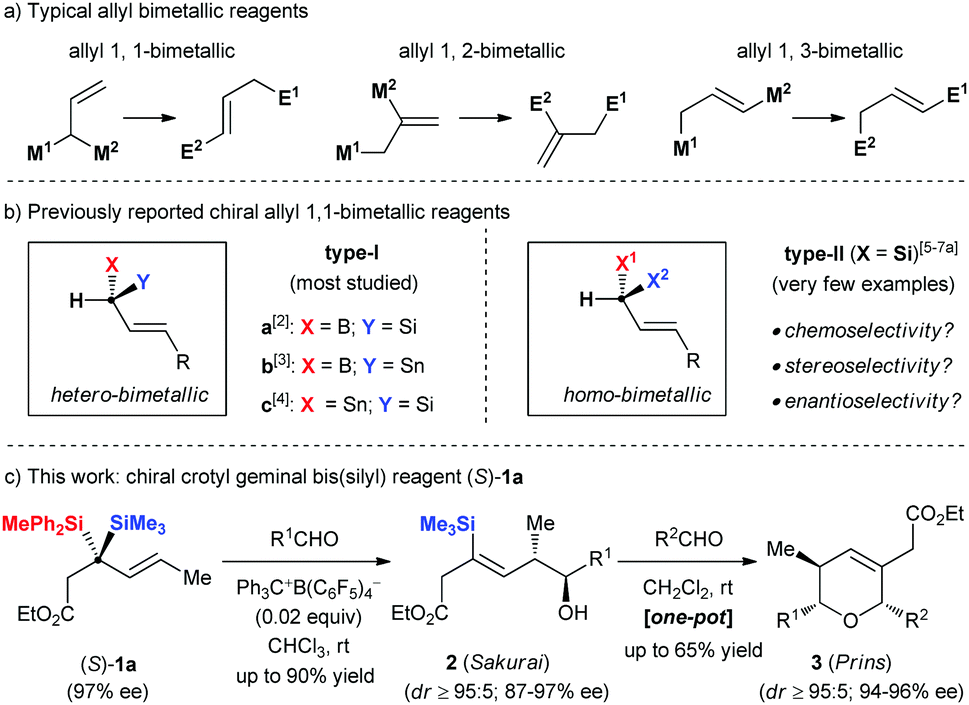
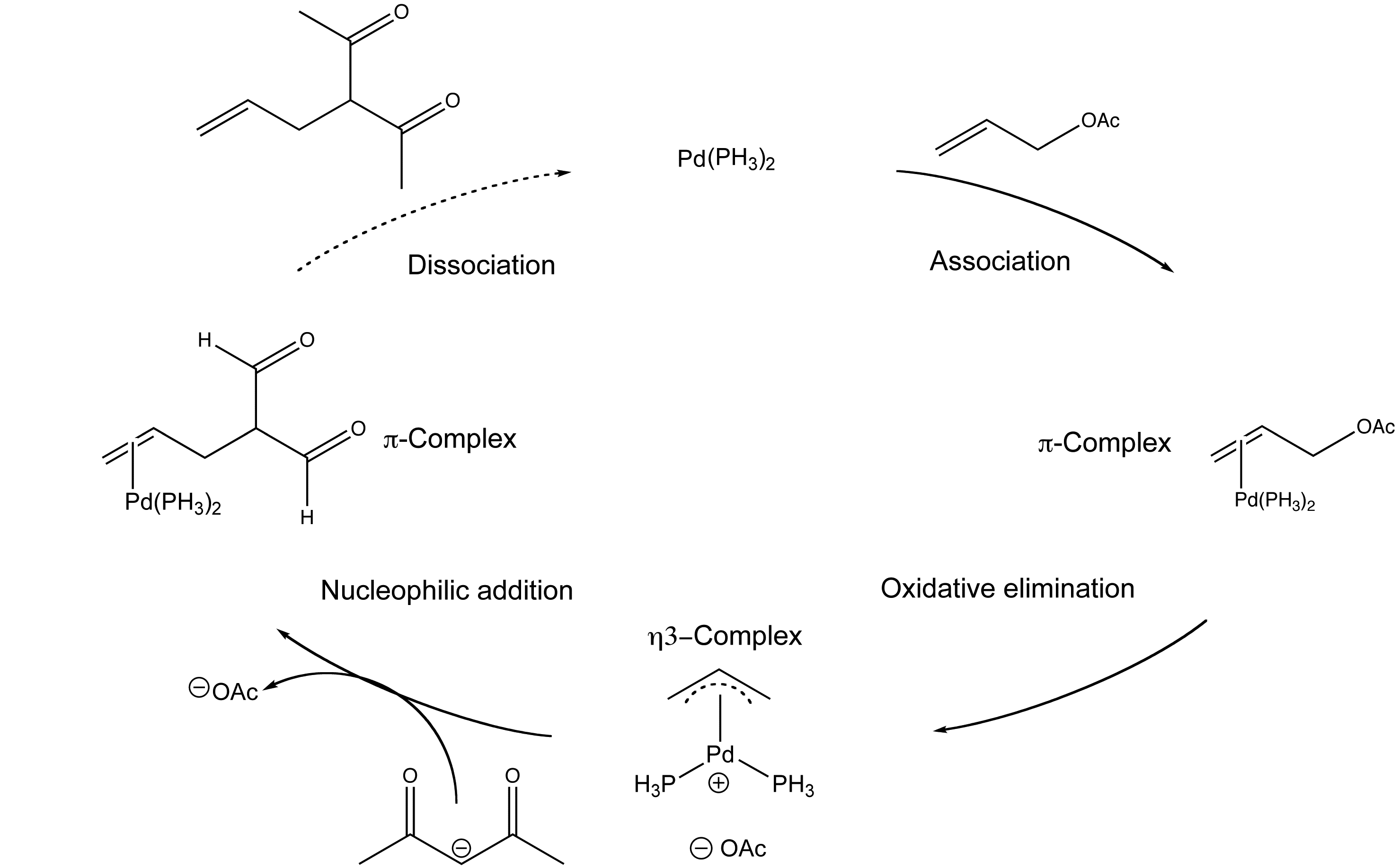
Alkene Vinyl Silane
An intermolecular anti markovnikov hydroamination of alkenes has been developed using triethyl phosphite and n hydroxyphthalimide.
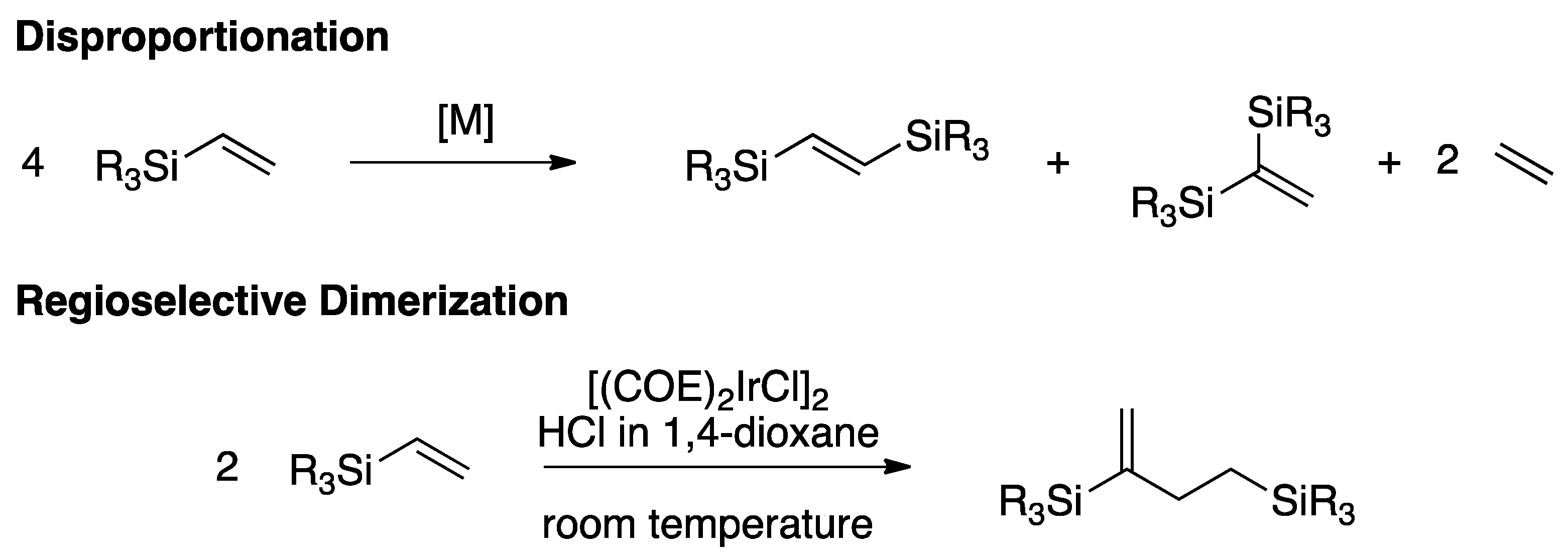
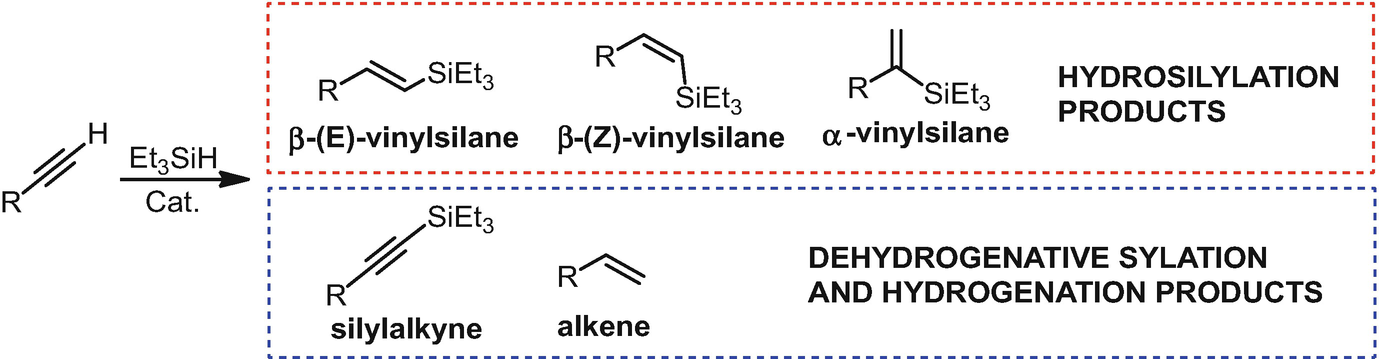
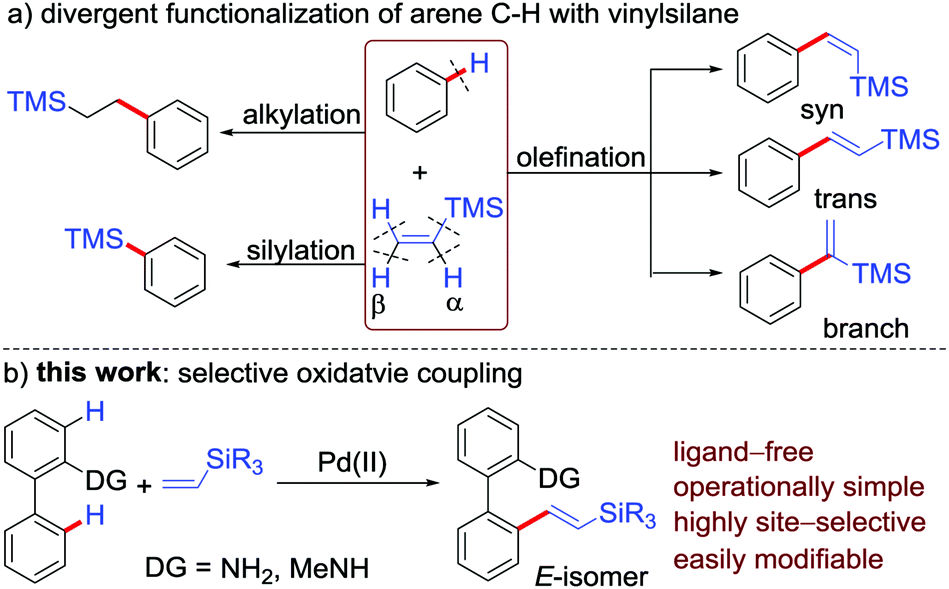
Alkene vinyl silane. Since then a number of studies have firmly established the mechanism of electrophilic attack on vinyl and allylsilanes the electron releasing strength of. The process tolerates a wide range of alkenes including vinyl ethers silanes and sulfides as well as electronically unbiased terminal and internal alkenes. 0 0 vinyl silanes react with electrophiles in a highly regioselective process in which the silicon is replaced by the electrophile at the ipso carbon atom. Six years later the generation of 2 iodoethyl trimethylsilane from vinylsilane and hi was observed.
In 1948 it was discovered that allyltrimethylsilane reacts with hydrobromic acid to give 2 bromopropyl trimethylsilane. Johnson synlett 2011 2811 2814. Adducts from the trichlorosilane reactions can be oxidised to afford alcohols in moderate yields. Terminal vinyl silanes heck coupling non alkyl si substituents are required to activate the vinyl silane to heck cross coupling subsequent functionalisation of the silyl diene alkoxysilanes generally require forcing conditions and even then react only in poor yields with significant protodesilylated products recovered.
Gaseous and flammable silanes are inconvenient to use in chemical reactions. The resultant n alkylphthalimides can readily be transformed to the corresponding primary amines. A copper catalzed functionalization of propiolate esters with various grignard reagents in presence of trimethylsilyl trifluoromethanesulfonate enables the synthesis of substituted e vinyl silanes in good yields and excellent diastereoselectivities via a catalytic carbocupration silicon group migration sequence.