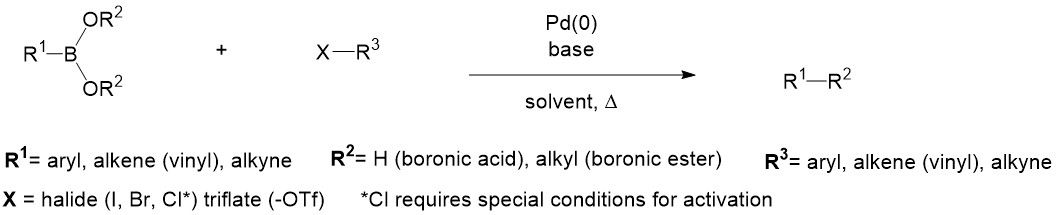
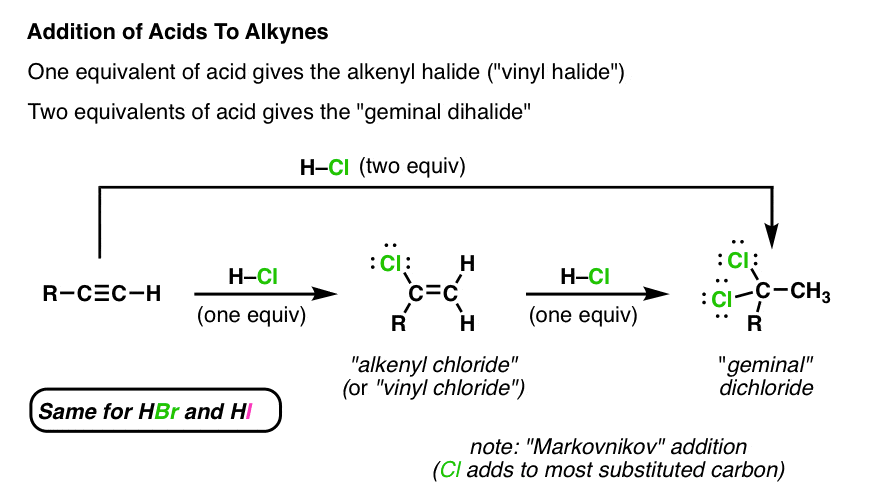Alkyl Aryl Vinyl Difference
Allyl groups have three carbon atoms and five hydrogen atoms.
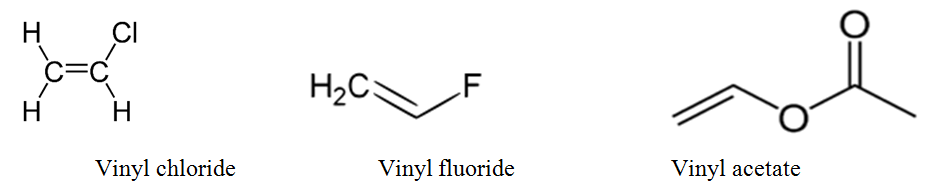
Alkyl aryl vinyl difference. Noun chemistry uncountable the univalent radical ch 2 ch derived from ethylene countable any of various compounds and substances containing the vinyl radical especially various tough flexible shiny plastics. Main difference alkyl vs aryl. However alkyl halides may sometimes be confused with aryl halides. A vinyl halide is clearly a species with a formula h 2c c x h in which a halide is directly bound to an olefinic bond formally this is ethylene h 2c ch 2 with one of the hydrogens substituted by a heteroatom vinyl chloride h 2c chcl is an example.
Vinylic carbocations are unstable as compare to the allyl carbocations as they lack p character. This is the main difference between allyl and vinyl groups. Key difference allyl vs vinyl both allyl and vinyl groups have slightly similar structures with a small variation. As nouns the difference between aryl and vinyl is that aryl is.
Both alkyl and aryl groups have carbon and hydrogen atoms. Difference between alkyl and aryl. Alkyl groups and aryl groups are two examples of functional groups. Vinyl contains two sp 2 hybridized carbon atoms and three hydrogen atoms.
An alkyl group is a functional group that can be. They are moieties of large molecules. Halogens are more electronegative than carbon. Vinyl indicates the ch ch 2 functional group which can be formed by removing hydrogen from ethylene.
For example if the halogen atom is attached to a carbon atom which is attached to a benzene ring cl ch 2 c 6 h 5 one would think it is an aryl halide but it is an alkyl halide because the halogen atom is attached to the carbon that is sp 3 hybridized. The main difference between alkyl and aryl groups is that alkyl groups do not have aromatic rings whereas aryl groups have aromatic rings in their structure. Alkyl anagrams vinyl. Functional groups are parts of organic molecules having the responsibility for the characteristic properties of a certain molecule.
The characteristics noted above lead us to anticipate certain types of reactions that are likely to occur with alkyl halides. In vinyl group c c is directly attached to the rest of the chain. The key difference between these two structural components is the number of carbon and hydrogen atoms. With the exception of hf pk a 3 2 all the hydrohalic acids are very strong small differences being in the direction hcl hbr hi.
In contrast to vinyl allyl group is attached to the rest of the molecule through ch 2 group. Alkyl and aryl are two chemical terms used to name functional groups of organic compounds. The main difference between the alkyl and the aryl is that alkyl is a kind of functional group which contains no aromatic ring whereas aryl is a kind of functional group which contains an aromatic ring.