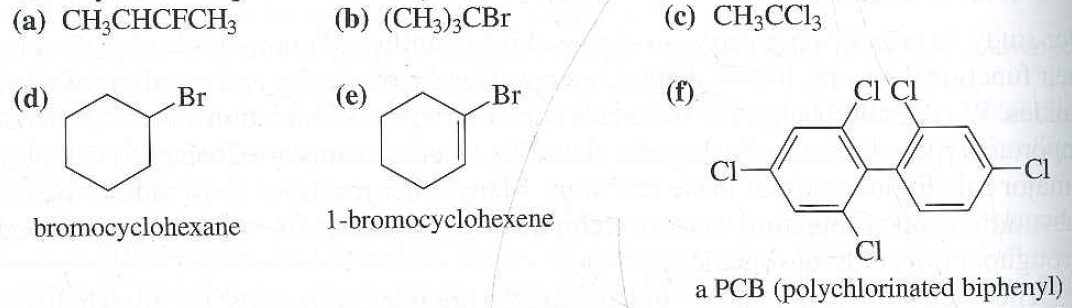
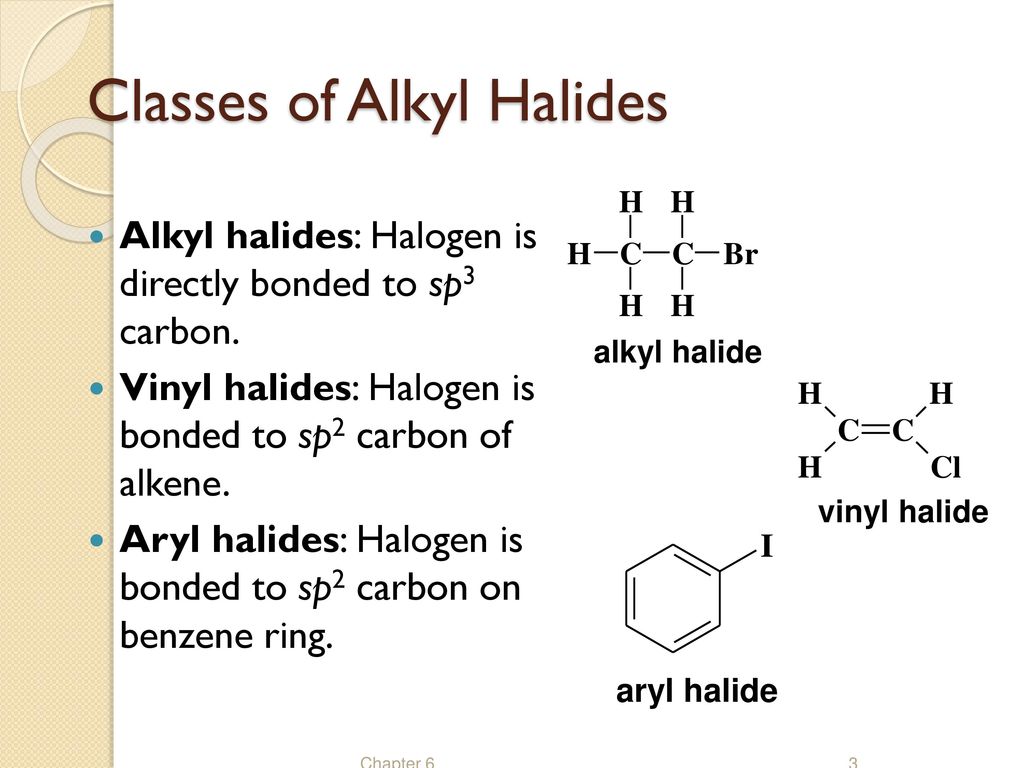
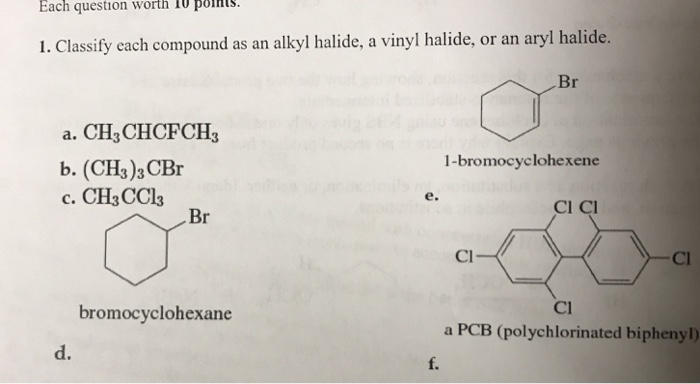
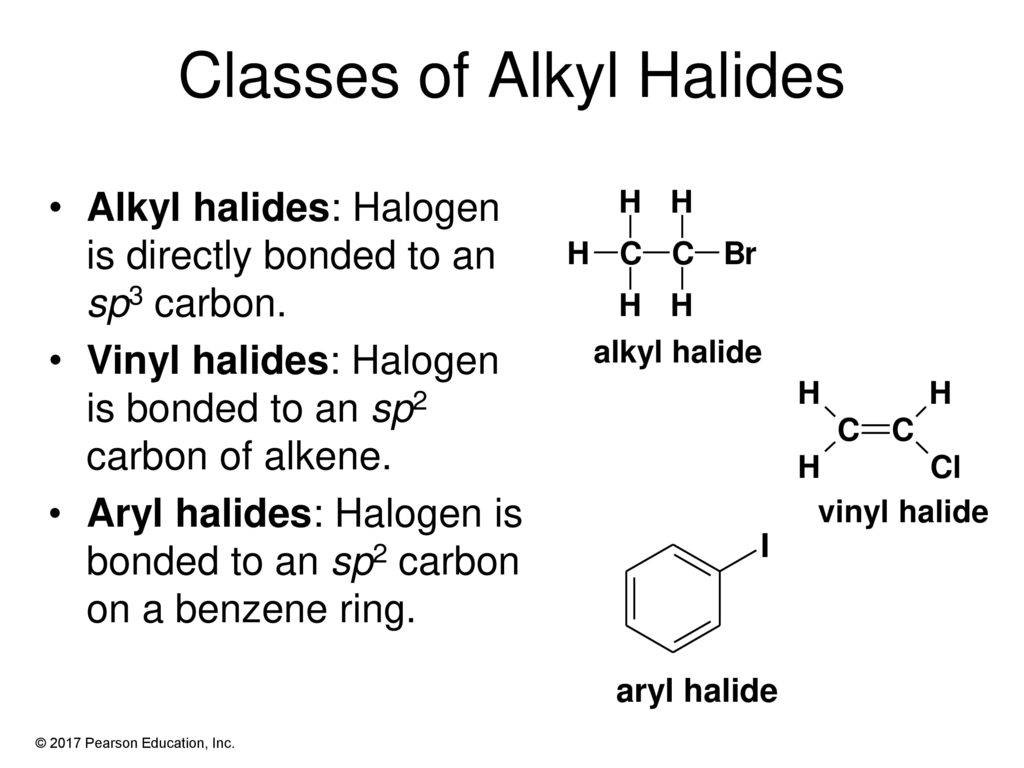
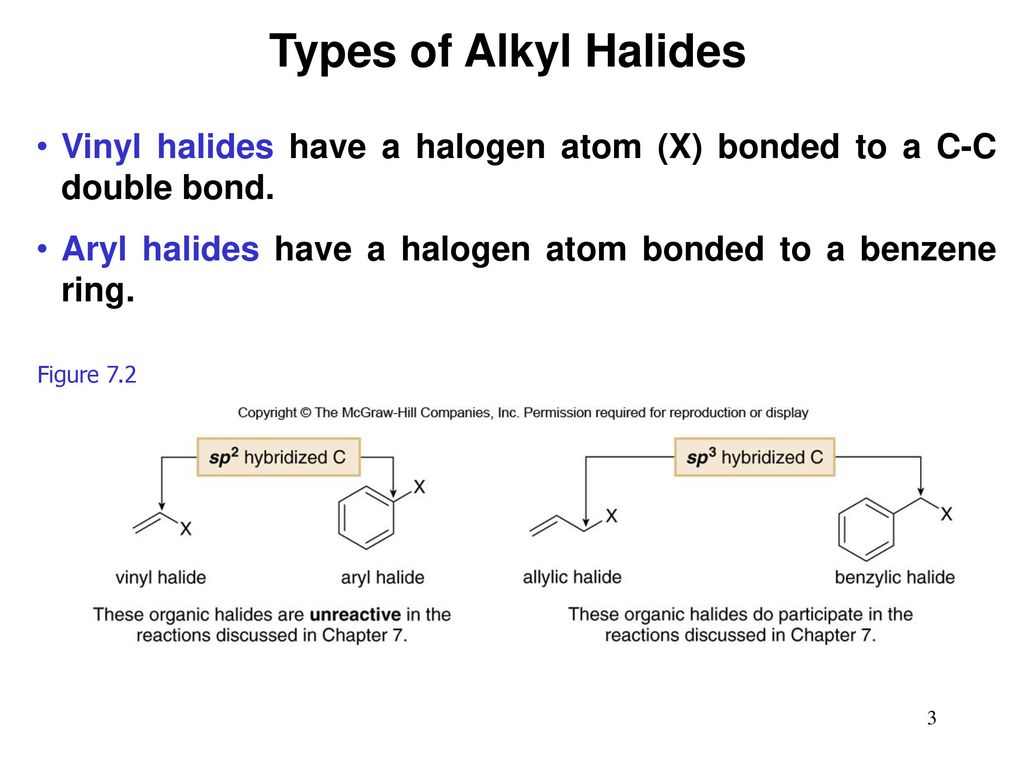
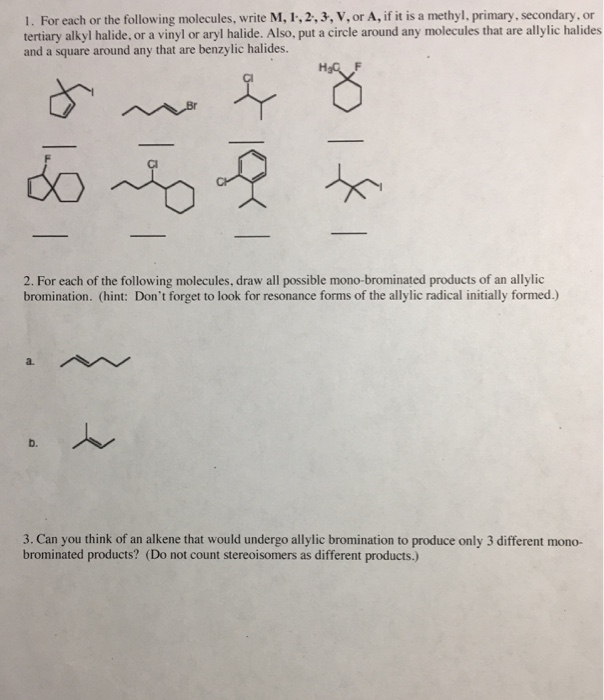
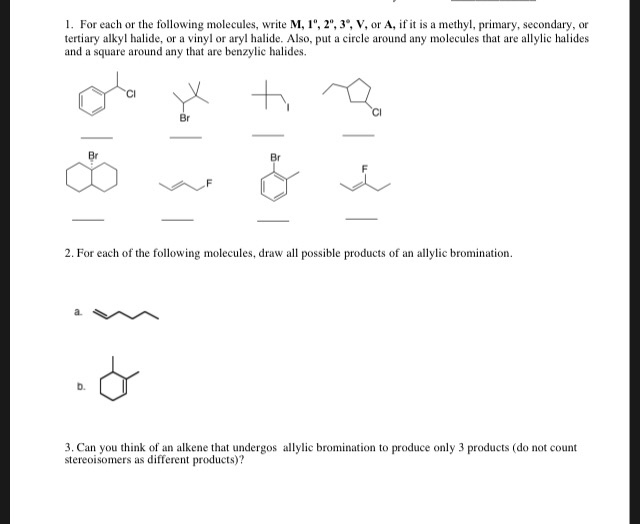
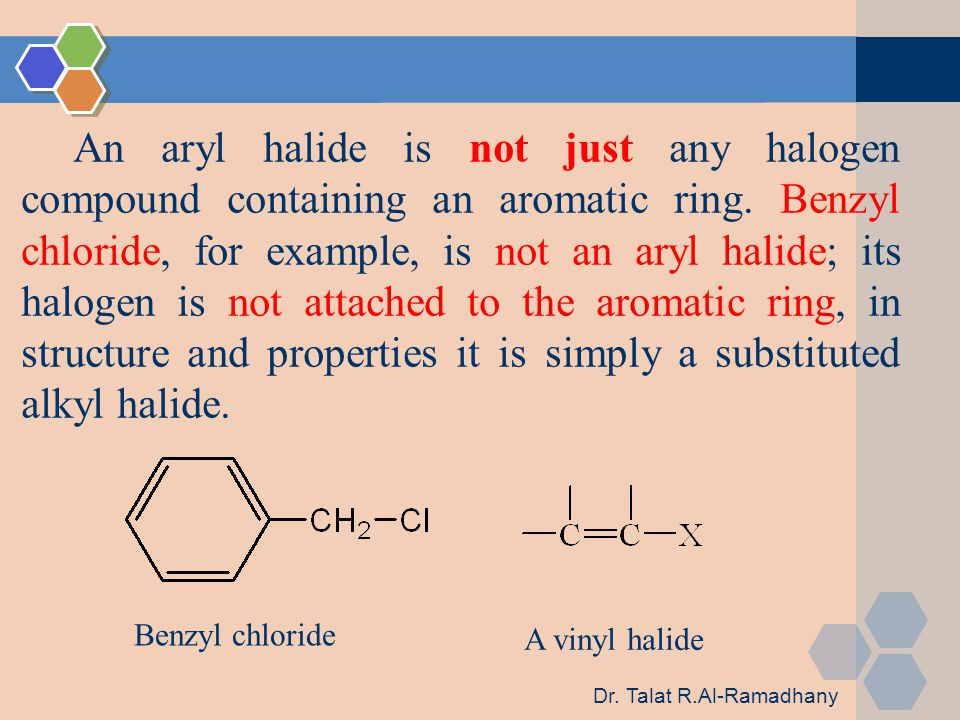
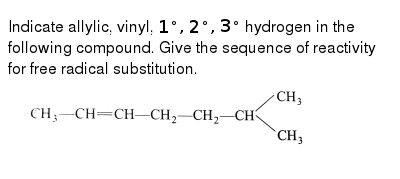
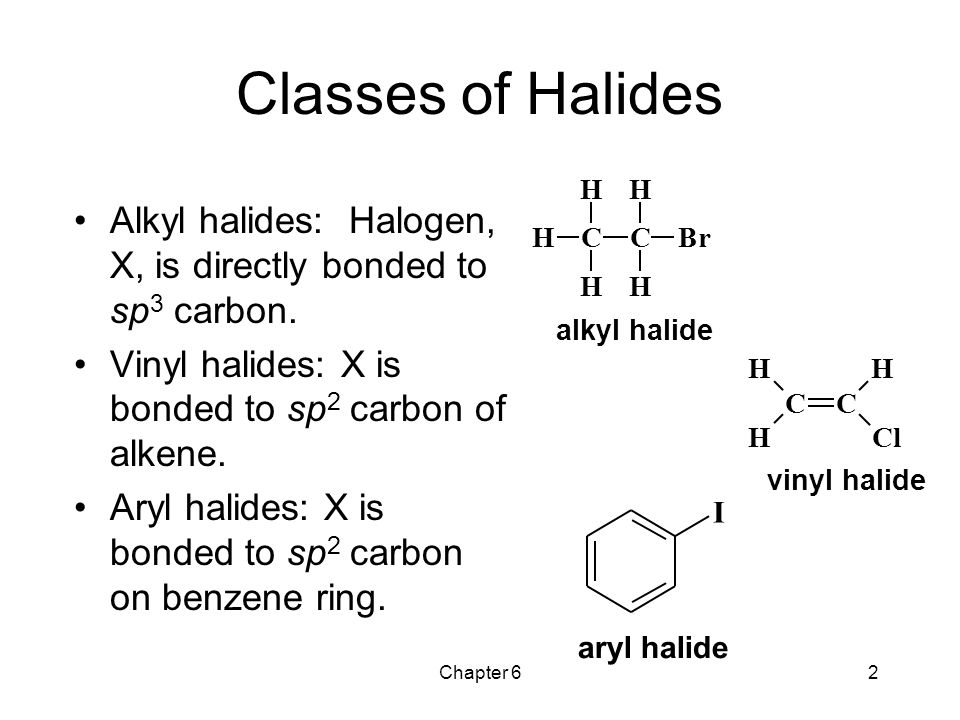
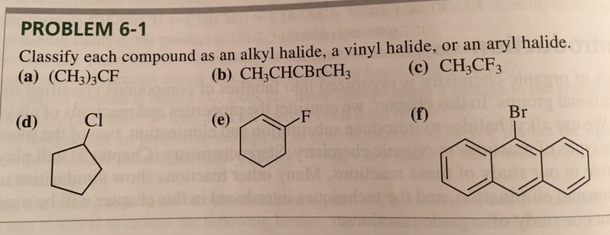Alkyl Vinyl Aryl Halides
There is an interaction between one of the lone pairs on the chlorine atom and the delocalized ring electrons and this strengthens the bond.
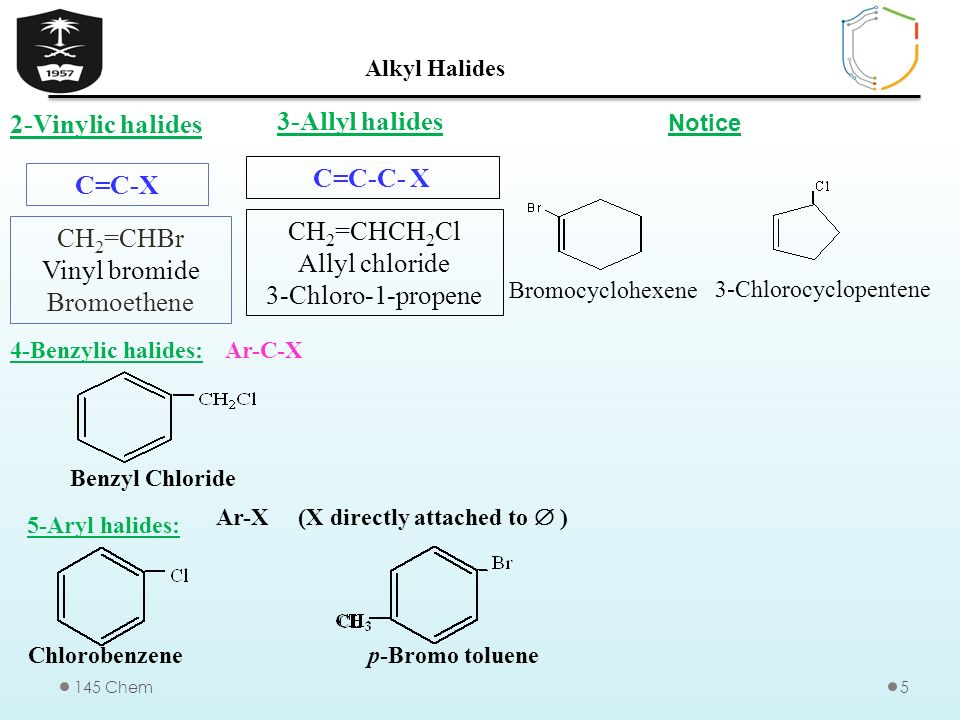
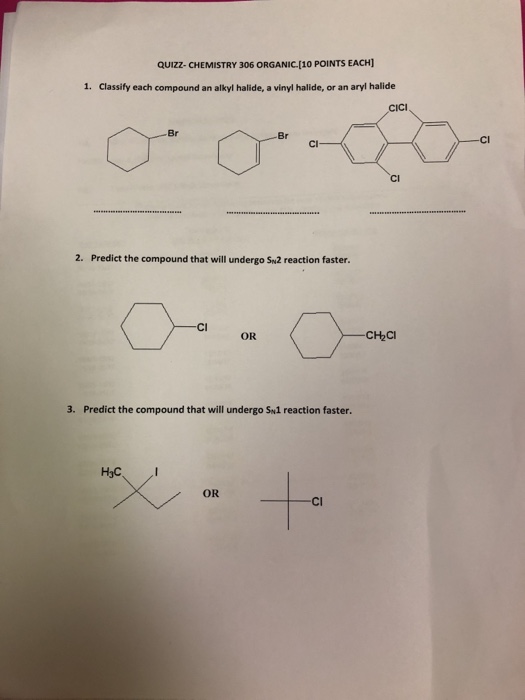
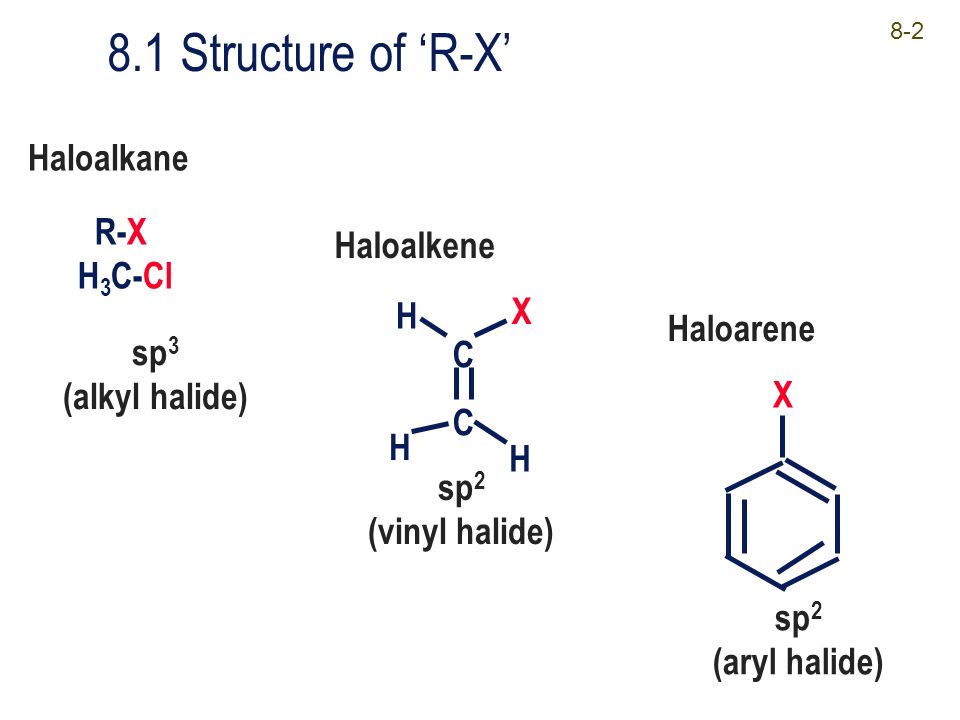
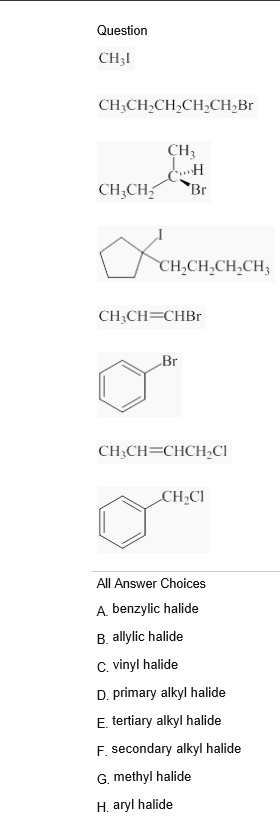
Alkyl vinyl aryl halides. Step 1 of 3 if the halogen atom halo group is bonded to a single bonded carbon atom a sp3 hybridized carbon atom then it is known as an alkyl halide. Unlike typical alkyl halides aryl halides do not participate in conventional s n 2 reactions as the backside attack required for an s n 2 reaction is impossible owing to the planar structure of the aryl group. The extra strength of the carbon halogen bond in aryl halides. For this reason alkenyl halides with the formula rch chx are sometimes called vinyl halides.
Benzylic halides allylic halides vinylic halides and aryl halides. A vinyl halide is clearly a species with a formula h 2c c x h in which a halide is directly bound to an olefinic bond. If the halogen atom halo group is bonded to a double bonded carbon a sp2 hybridized carbon atom then it is known as vinyl halide. The carbon chlorine bond in chlorobenzene is stronger than you might expect.
An aryl halide has general formula c6h 5x in which an halide group x has substituted the aryl ring. Vinyl chloride h 2c ch cl is an example. Consequently this functional group is polarized so that the carbon is electrophilic and the halogen is nucleophilic as shown in the drawing below. Formally this is ethylene h 2c ch 2 with one of the hydrogens substituted by a heteroatom.
The functional group of alkyl halides is a carbon halogen bond the common halogens being fluorine chlorine bromine and iodine. In organic chemistry a vinyl halide is a compound with the formula ch 2 chx x halide.