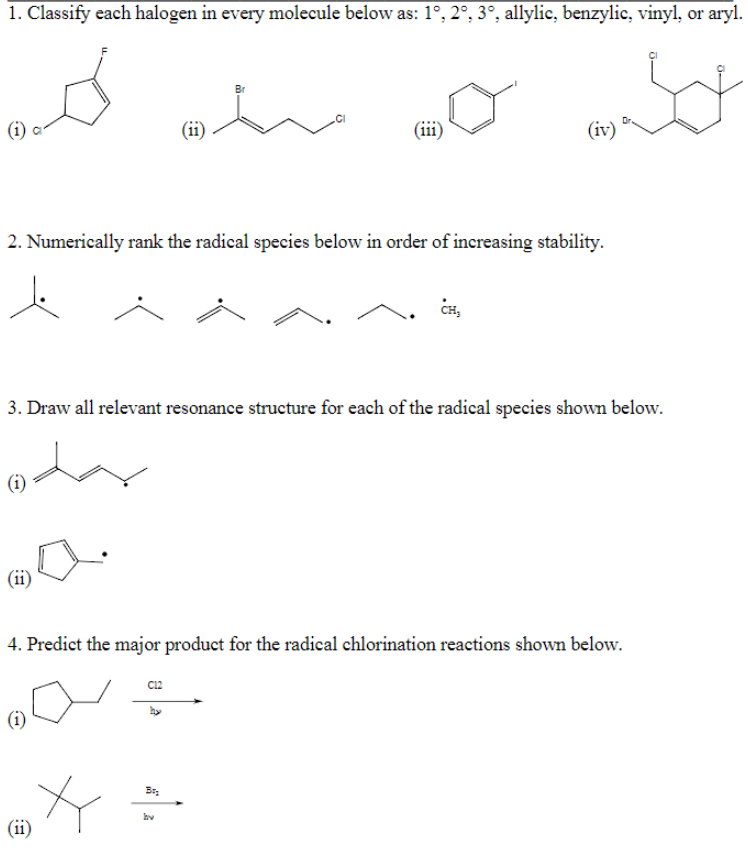
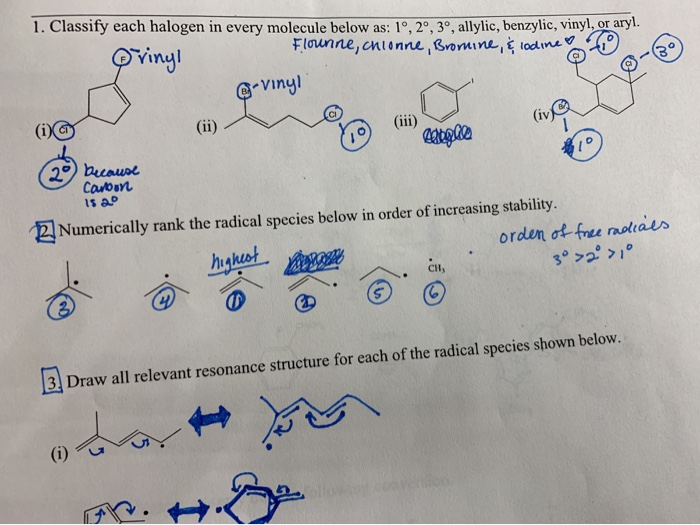
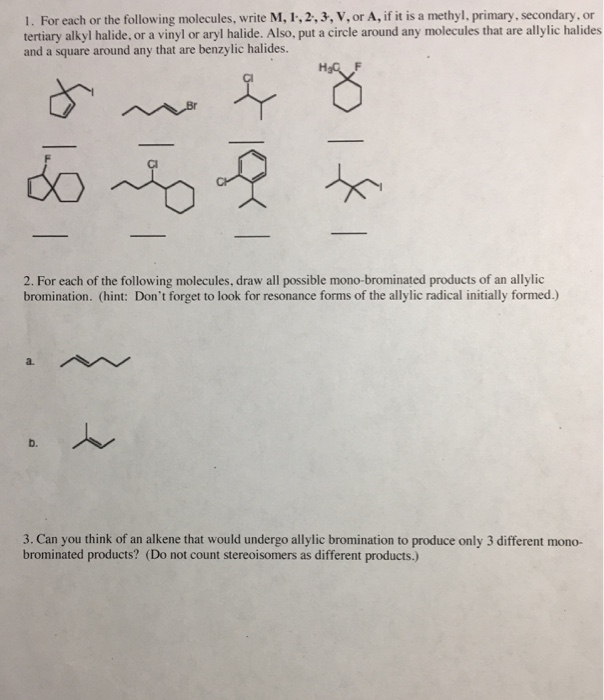
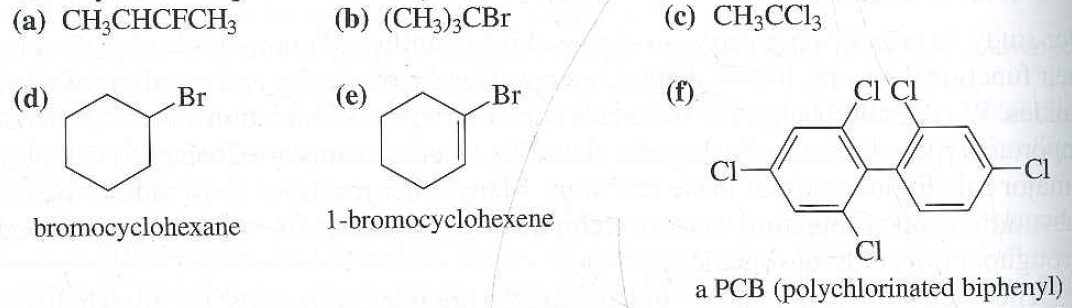
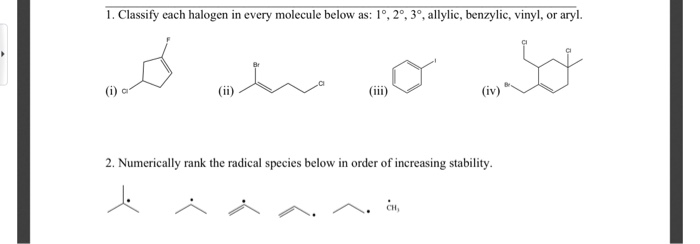
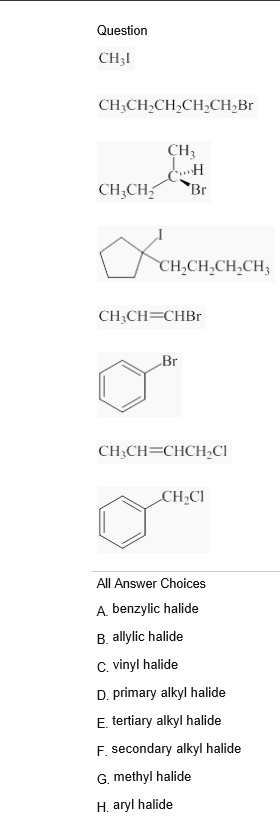
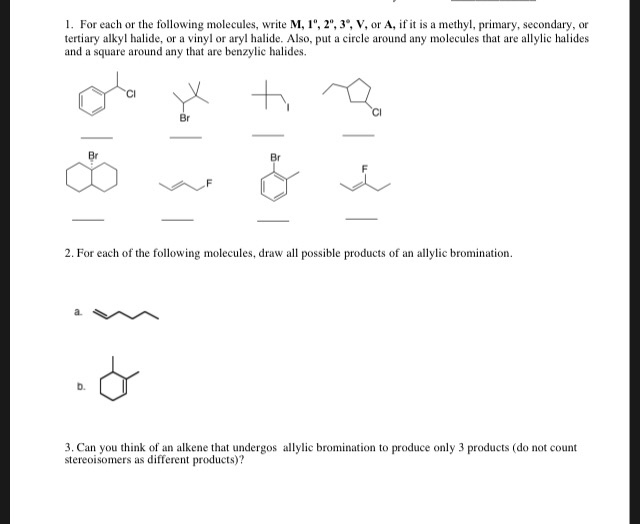
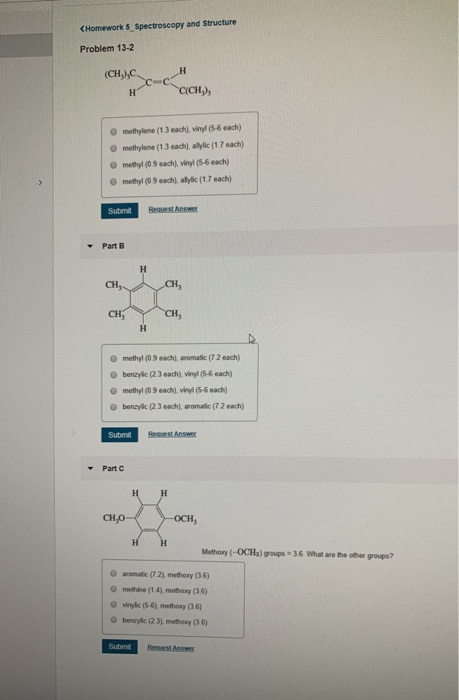
Allylic Vs Benzylic Or Vinylic
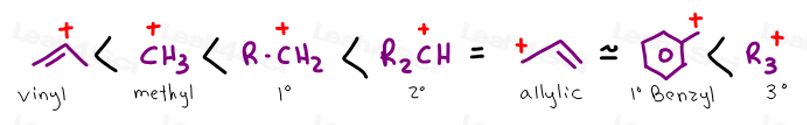
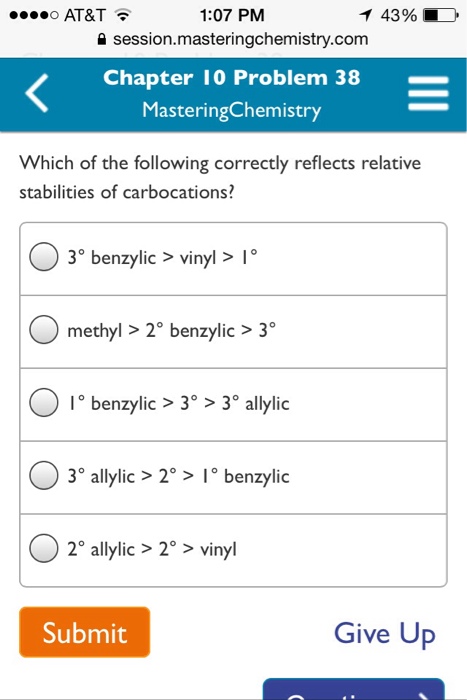
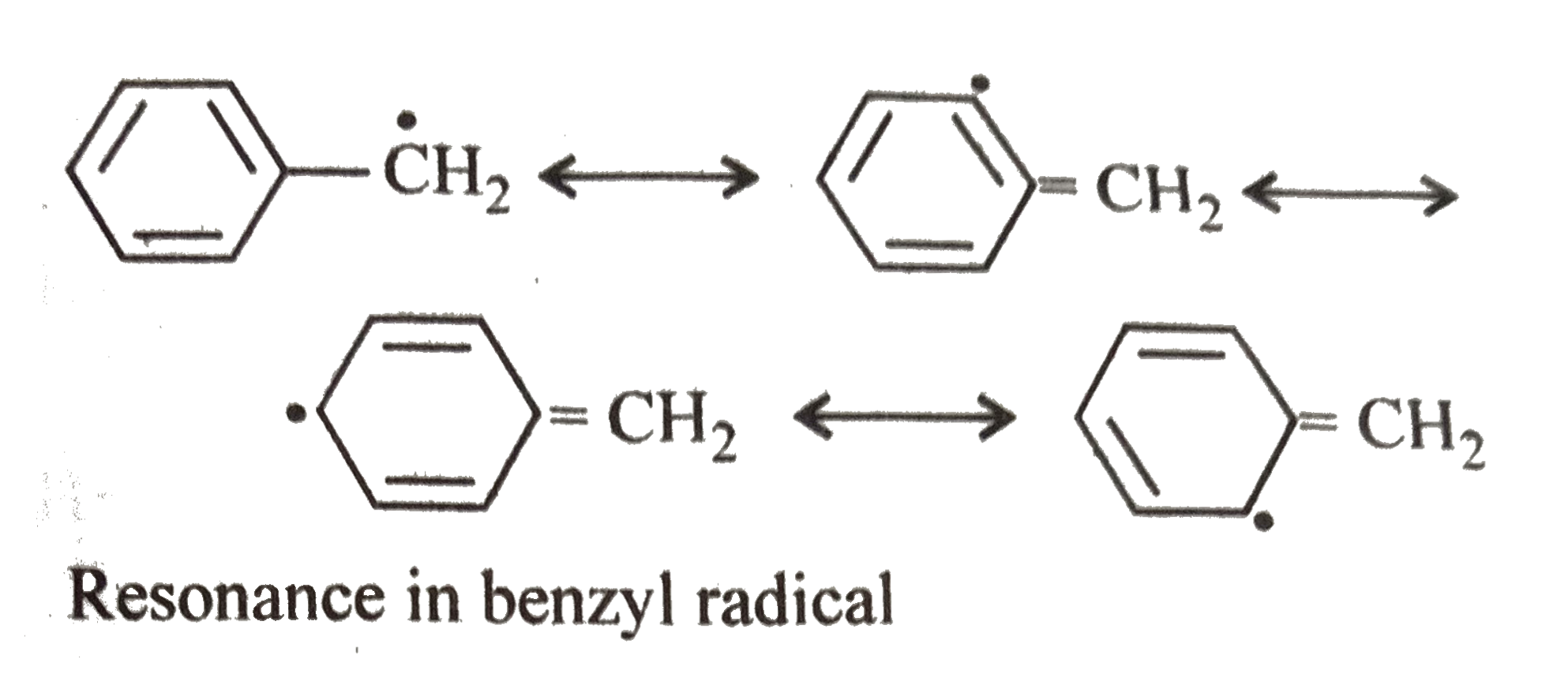
Allyl form a stable carbocation because of the electron delocalization.
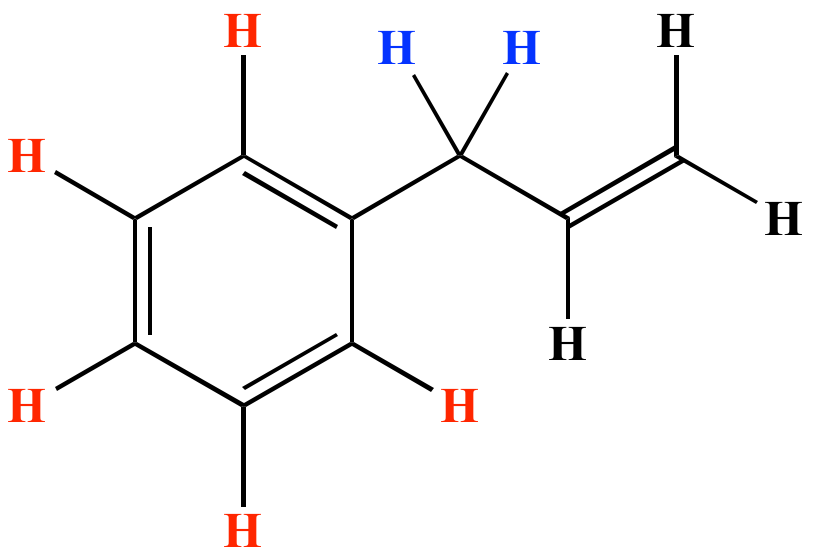
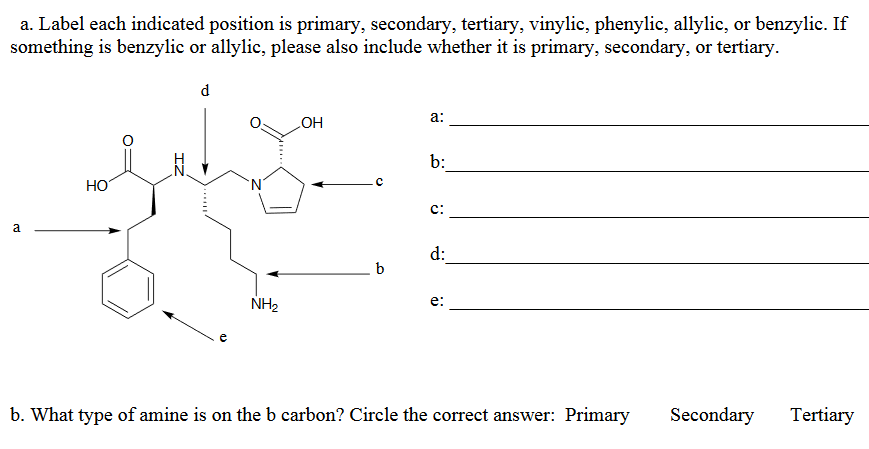
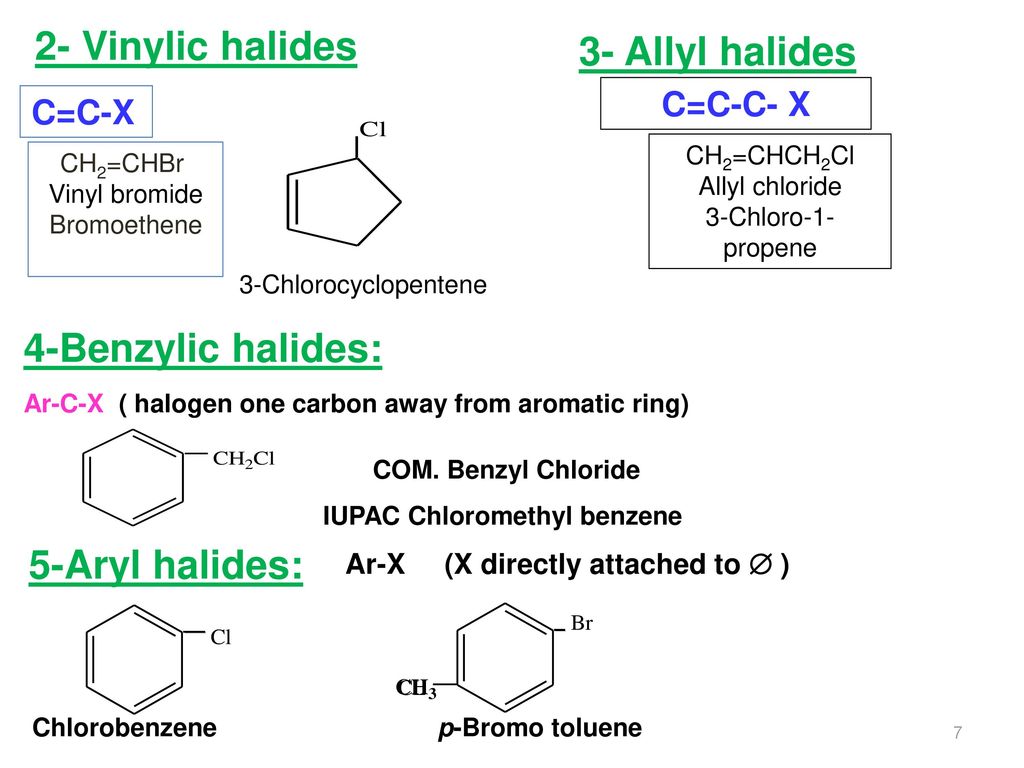
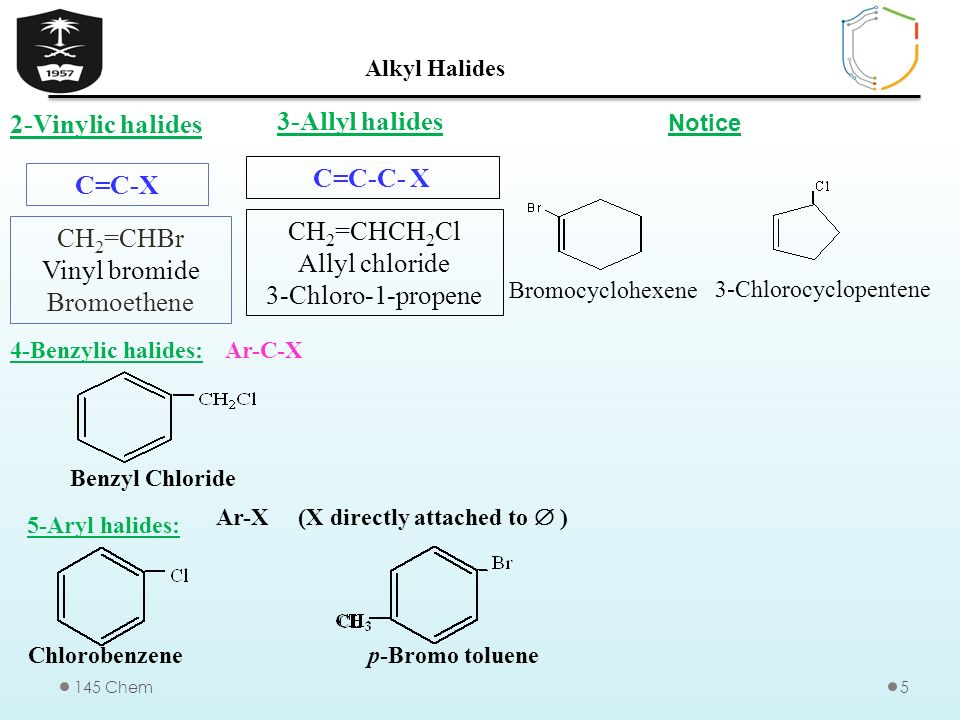
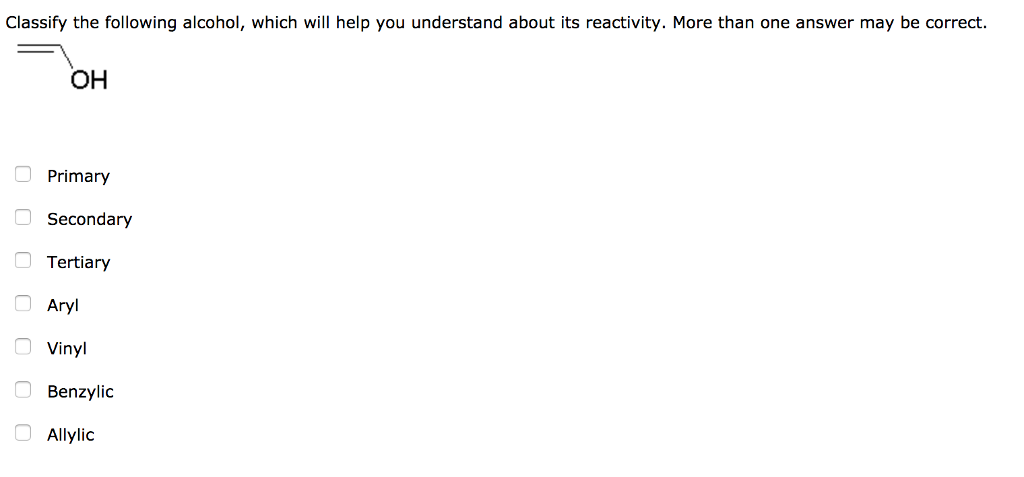
Allylic vs benzylic or vinylic. None of the other hydrogens are vinylic. Key difference allylic vs vinylic carbons functional groups are very important in understanding the different physical and chemical properties of organic molecules the terms allylic and vinyl carbons indicate whether the carbon atom is bonded directly or indirectly to a double bond in a molecule. I have heard about allylic vinylic benzylic carbons but positions. Vinyl group has two carbon atoms and three hydrogen atoms.
Allyl group holds three carbon atoms and five hydrogen atoms. Vinylic carbocations are unstable as they lack p character. Key difference allyl vs vinyl both allyl and vinyl groups have slightly similar structures with a small variation. Rapid s n 2 substitution for 1º.
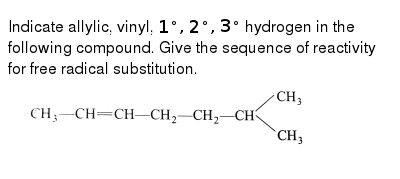
An allylic hydrogen is a hydrogen atom that is bonded to an allylic carbon in an organic molecule. The vinylic hydrogens are shown in red. For 3º halides a very slow s n 2 substitution or if the nucleophile is moderately basic e2 elimination. The key difference between these two structural components is the number of carbon and hydrogen atoms.
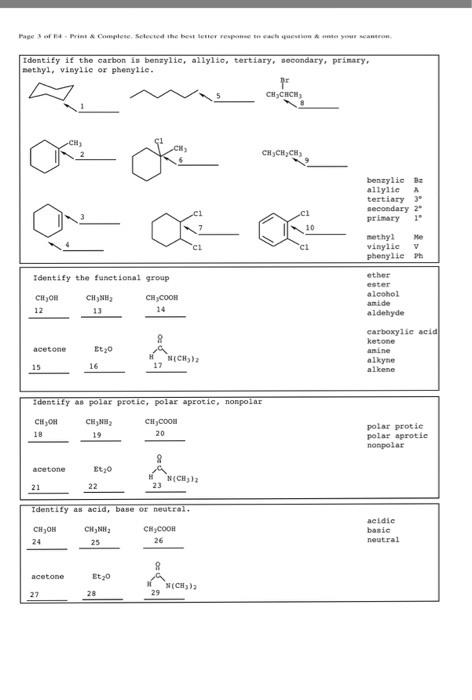
The key difference between allylic and vinylic carbon is that allylic carbon is the carbon. In high dielectric ionizing solvents such as water dimethyl sulfoxide acetonitrile s n 1 and e1 products may be observed. Allyl groups have three carbon atoms and five hydrogen atoms. Benzylic position allylic position propargylic position aryl aryl hydrogen methyl hydrogen primary hydrogen secondary hydrogen tertiary.
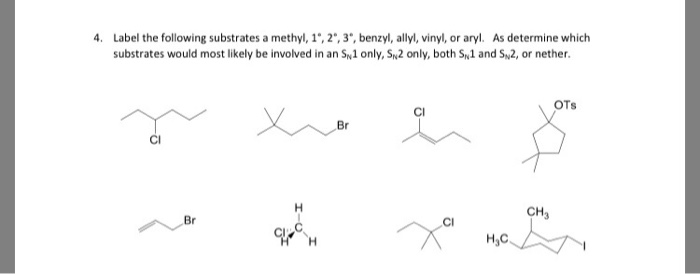
A π bonds only at allylic position b π bonds only at vinylic position c π bonds at allylic and benzylic position also d i effect while the answer is obviously not d i am really confused about what allylic vinylic and benzylic positions actually mean. Vinyl group vinylic hydrogen vinylic carbocation allylic position benzylic position propargylic position wikipedia entry return to glossary index. But would the c bound to the 2 carbons in a double bond also be considered an allylic carbon. Allyl group gets attached to any other group of atoms through.
Allyl h 2 c chch 2 rapid s n 2 substitution for 1º and 2º halides. Benzene c x where x is the benzylic group vinylic. Both groups own a double bond between two carbon atoms where all the other atoms are bonded through single bonds. Carbo cations may be stabilized by.